Castor’s AI platform deploys studies in under 4 weeks, cuts data entry time by 80%, and resolves queries 92% faster. Human-in-the-loop oversight at every step.
The delays are predictable. Most of them are preventable.
Protocol-to-first-patient timelines stretch because UAT is manual, translation reviews are sequential, and test environment configuration requires weeks of back-and-forth. The industry standard is 12-16 weeks of setup before a single patient is enrolled.
Extracting regulatory-grade variables from unstructured records for FDA submissions, 510(k) filings, and external control arms requires large teams working for months. Every chart review study carries this cost by default.
Errors found at the end of a study take weeks to resolve. Most sites batch their data review, so discrepancies that could have been caught at entry compound into query backlogs that push lock dates by weeks or months.
AI in clinical trials is more than workflow automation. Agentic AI plans, acts, and adapts across the entire trial lifecycle. It reads protocols, configures environments, extracts data from unstructured records, and flags anomalies in real time.
Castor uses AI in three distinct ways: accelerating study build and UAT, automating intelligent data extraction and validation, and detecting data errors before they become query backlogs. The key principle across all three is human-in-the-loop oversight at every critical decision point.
1
2
3
4
Three integrated capabilities, from protocol receipt to database lock.
AI Time Travel UAT interprets your protocol and auto-configures test environments with historical timestamps. Concurrent AI-assisted translations in 20+ languages eliminate the multi-week localization delays that stretch conventional setup timelines. More than 90% of Castor studies are live in under 4 weeks.
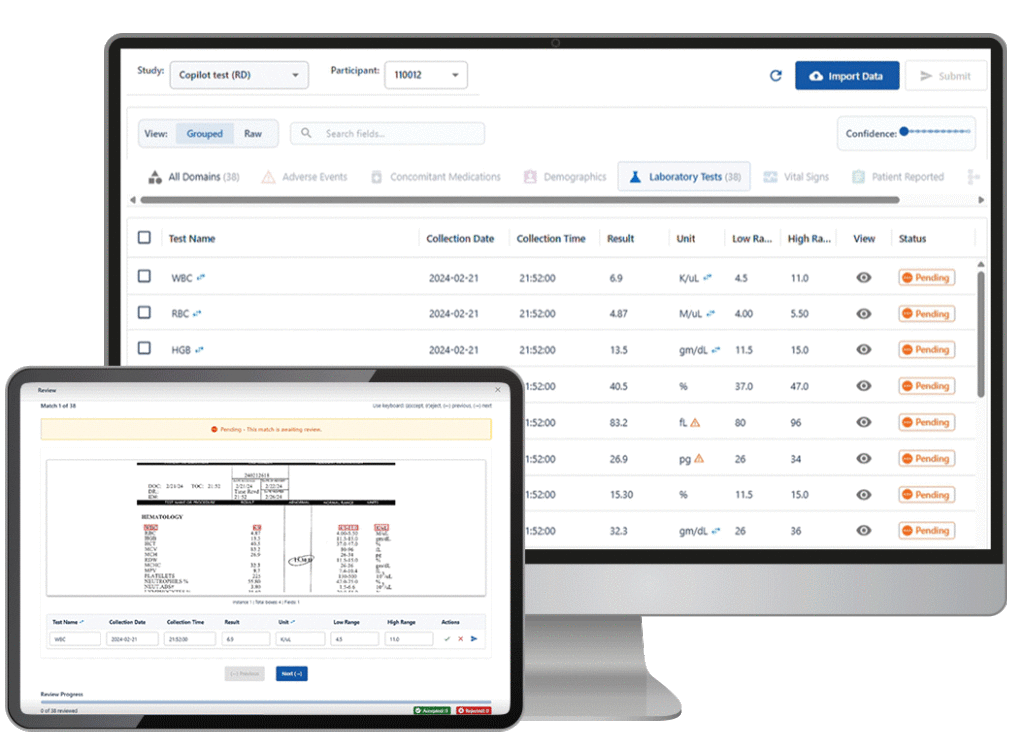
Castor Catalyst reads unstructured records from EMRs, PDFs, and wearables via HL7 FHIR-compliant connections. It extracts regulatory-grade variables, maps data to CDISC SDTM/ADaM, and pushes validated entries to your electronic data capture system. Post-validation EDC error rate is 0%.
AI anomaly detection flags discrepancies at the point of data entry, before they compound into end-of-study query backlogs. Queries surface and close in real time. In a verified 12-country HEOR study, this approach delivered database lock and define.xml in under 2 weeks.
Here is how Castor automates the journey from patient recruitment to submission-ready RWE data, while ensuring quality through rigorous human validation and risk-based quality management9.
Castor connects to EMRs, claims databases, wearables, and site data via FHIR API or upload. Every source is logged with a timestamp and linked to any downstream output.
The AI reads each input, executes the relevant workflow (extraction, anomaly check, translation review), and assigns a confidence score. High-confidence outputs proceed automatically. Everything else goes to review.
3
Outputs below the configurable confidence threshold go to a qualified reviewer. They see the source alongside the AI output, then approve or correct before anything commits to the trial record.
4
Validated output commits to your EDC with a complete ALCOA+ compliant audit trail. Query volume stays low throughout the study. Database lock in under 2 weeks.
| Metric | Castor AI | Manual process | Point solutions |
|---|---|---|---|
| Study deployment | Under 4 weeks | 12-16 weeks | Varies |
| Data entry time | 80% reduction | Full manual effort | Partial |
| EDC error rate | 0% post-validation | 8.54% baseline | Varies |
| Query resolution | 92% faster | 4-12 week cycles | Varies |
| Database lock | Under 2 weeks | Months | N/A |
Three common profiles. One platform.
You need to meet EU MDR or PMCF requirements, or gather FDA 510(k) registry data, without the abstraction budgets available to large pharma sponsors.
Castor Catalyst extracts regulatory-grade variables directly from site EMRs and PDFs, lowering your abstraction costs by up to 70%. Real-world evidence generation starts faster, at a fraction of conventional cost.
You’re running Phase II/III trials with a lean internal team and can’t afford UAT delays, endless query cycles, or a database lock that slips by months.
AI Time Travel UAT gets your study live in under 4 weeks. Real-time anomaly detection keeps you lock-ready throughout the trial. Your lean team stays focused on science, not administration. Built for biotech clinical trials.
You’re bidding on complex RWE or chart review studies and need a competitive edge to win sponsor business while protecting your margins.
Offer sponsors faster database locks and more cost-effective SDV. Your team uses Castor to abstract data at scale, validating AI output and delivering cleaner datasets in less time. Win more bids. Deliver on them.
Verified outcomes from production studies.
FHIR-based API integration enabled automated abstraction from 1,500+ patient records. The study reduced manual transcription by 90%, cut total study cost by 80%, and completed 70% faster than projected timelines. Part of a broader real-world evidence program.
Full enrollment and EDC data entry for 101 subjects completed over a single weekend. Zero query backlog at database lock. The study demonstrated how Castor’s biotech clinical trials platform handles concentrated enrollment windows without slowing data quality.
Castor’s EDC and AI data management infrastructure supported the evidence generation required for FDA Breakthrough Device designation and subsequent Class II listing. The electronic data capture system provided the audit trail foundation for FDA review.
Castor-supported trial data contributed to FDA Class II device listing and commercial insurance reimbursement approval. The eCOA solutions and AI-driven data management delivered a submission-ready dataset at every regulatory milestone.
“What drew me to Castor was the ease of use, and the ability to customize it easily to our setting in our clinical trial. Ease of use, adaptability, and reliability are three things that are really crucial in all these platforms.”
“Castor has been the most user-friendly platform I have used for data entry. It is clear and succinct and is easy to navigate. It allows me to enter data and respond quicker to queries.”
Agentic AI in clinical trials is a system that autonomously plans, acts, and adapts across the trial lifecycle: accelerating study build and UAT, extracting regulatory-grade variables from unstructured patient records, and detecting data anomalies in real time to accelerate database lock. Castor’s AI platform uses mandatory human-in-the-loop sign-off at every critical decision point before data commits to the clinical database.
In Castor’s AI platform, every automated workflow includes a mandatory human review stage. For data extraction via Castor Catalyst, the AI assigns a confidence score to each data point. Records below the configurable threshold are flagged for review by medically trained clinical staff before committing to the EDC. This approach produced a 0% EDC error rate post-validation, compared to an 8.54% pre-validation baseline. For study build workflows, AI-generated configurations and translations are reviewed by qualified study designers before activation.
Castor deploys studies in under 4 weeks. The industry standard for study build and UAT is 12-16 weeks. Castor’s AI Time Travel UAT testing interprets the study protocol, automatically configures test environments with historical timestamps, and resolves test scenarios without the manual back-and-forth that extends traditional timelines. AI-assisted translations in 20 or more languages run concurrently rather than sequentially, eliminating multi-week localization delays. More than 90% of Castor studies are live in under 4 weeks.
Castor’s real-time anomaly detection flags data discrepancies at the point of data entry, not after a weekly batch review cycle. When a site coordinator enters a value outside normal range or inconsistent with prior entries, the AI surfaces the issue immediately. This keeps query volume low throughout the trial and produces a 92% reduction in query resolution time compared to conventional end-of-study query cycles. In a verified 12-country HEOR study, this approach enabled database lock and define.xml delivery in under 2 weeks. For teams managing decentralized clinical trials, point-of-entry detection is especially critical given the distributed site structure.