
Categories
Connect with us on LinkedIn
Beyond the EHR: meeting the FDA’s new real-world evidence standards
The FDA’s December 2025 device guidance and March 2026 ICH M14 adoption raise the bar for RWE programs....

Phase 4 and real-world evidence: not a spectrum, a strategic choice
Phase 4 is a post-approval interventional trial that fulfills regulatory commitments. Real-world evidence spans PASS, PAES, HEOR, natural...
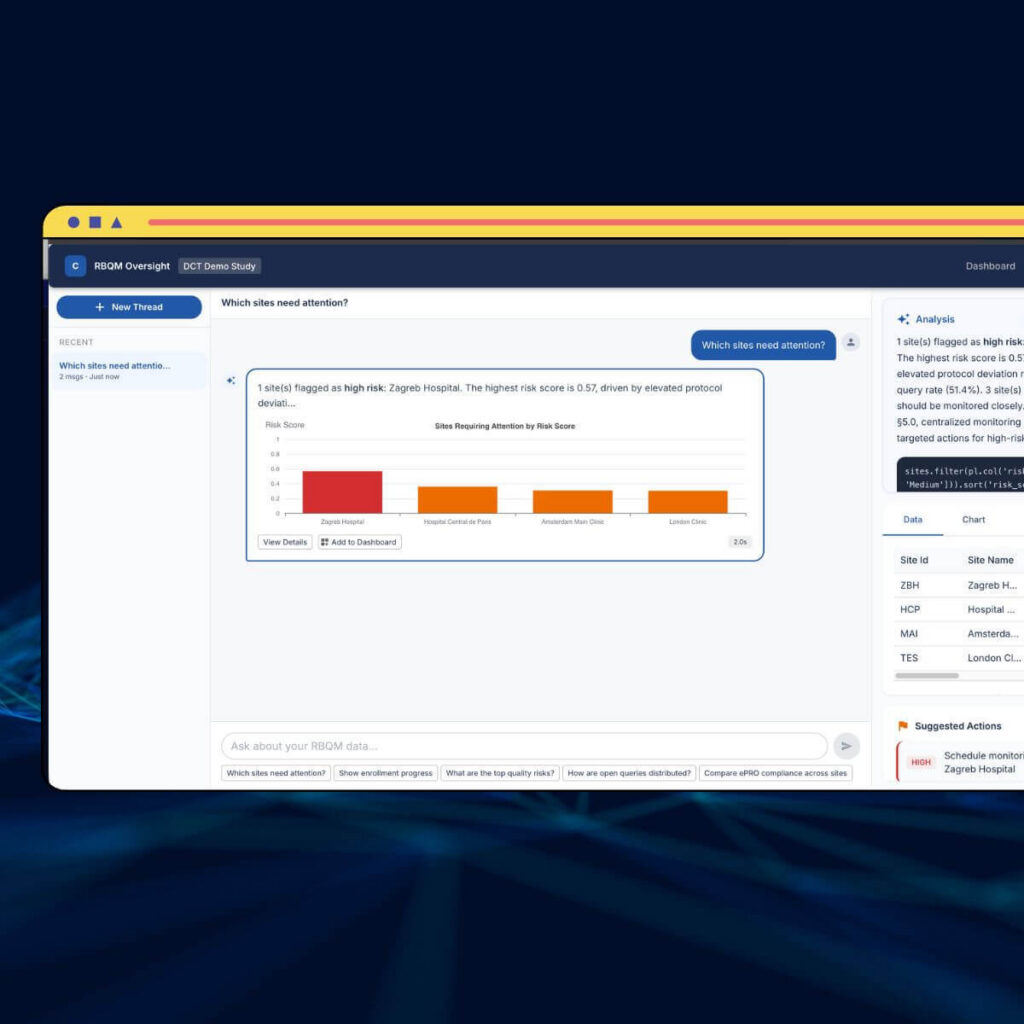
ICH E6(R3) is here: what your centralized monitoring strategy needs right now
ICH E6(R3) is in effect. Castor's recap covers centralized monitoring, sponsor accountability, and closing the biotech RBQM gap.
How a 3-Person Team Got to Phase 3 — Without the Overhead
Gameto proves you don’t need a large CRO or a floor of data managers to run complex global...

From Hype to Health: What Sword Health Got Right About Evidence
Sword Health didn’t follow the typical digital health playbook. Instead of rushing to market, they invested early in...

Unlocking FDA’s Vision for PRO Collection in RWE: Timing, Methods, and Insights
Discover FDA's guidance on collecting PROs for Real-World Evidence (RWE), focusing on timing, methods, and improving data quality.

Understanding Phase 4 Clinical Trials: A Critical Component of Post-Marketing Insight to Drugs in the Real-World
When it comes to Phase 4 clinical trials, Castor's cutting-edge Real-World Evidence (RWE) platform is your key to...

5 Key Takeaways from the FDA’s Real-World Evidence Guidance
A deep dive into the evolving regulatory environment, the innovative approaches emerging in RWE, and the critical importance...

Navigating the Evolving Regulatory and Data Landscape
A deep dive into the evolving regulatory environment, the innovative approaches emerging in RWE, and the critical importance...

Revolutionizing Blood Pressure Monitoring: Insights from Aktiia and Castor’s Collaboration
How Aktiia brought their continuous BP device to market by integrating vast amounts of blood pressure data with...

Measuring the true patient experience in Cancer Trials with eCOA / ePRO
Shedding light on the complexities and advancements in measuring PROs, drug tolerability, and quality of life in cancer...

Unlocking the power of Real-World Evidence in FDA submissions
An avalanche of data from so many real-world sources is available today, with the promise of more data...