
Categories
Connect with us on LinkedIn
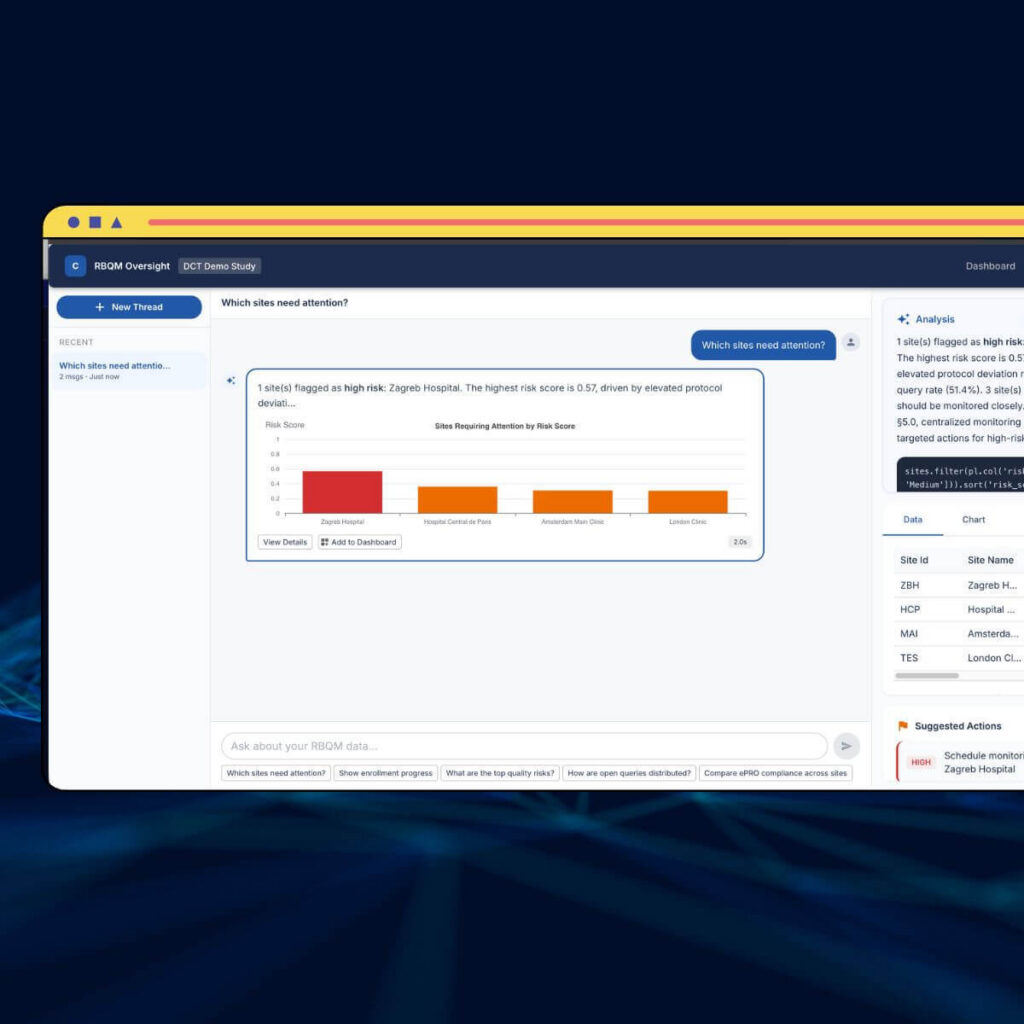
ICH E6(R3) is here: what your centralized monitoring strategy needs right now
ICH E6(R3) is in effect. Castor's recap covers centralized monitoring, sponsor accountability, and closing the biotech RBQM gap.
How a 3-Person Team Got to Phase 3 — Without the Overhead
Gameto proves you don’t need a large CRO or a floor of data managers to run complex global...

From Hype to Health: What Sword Health Got Right About Evidence
Sword Health didn’t follow the typical digital health playbook. Instead of rushing to market, they invested early in...

Building a Safer, Smarter Future: A Tiered Approach to AI in Life Sciences
Adopt AI in clinical research with confidence using a risk-based framework that ensures privacy, security, and regulatory alignment.

Why a Life Sciences Museum Matters—And What It Can Teach Us About the Future of Medicine
What a Museum of Medicine can teach us about trials, trust, and how the public sees (or doesn’t...

Build vs. Buy vs. Partner in Life Sciences: Making the Right Call
How do you decide between building, buying, or partnering? Learn key decision factors and red flags to avoid...

Do CROs Need to Reinvent Themselves in 2025? A Deep Dive with Greg Licholai and Derk Arts
High R&D costs aren’t due to a lack of innovation but inefficiencies in trials. Here’s how we can...

Local Labs in the EDC: Streamlining Safety and Efficacy Insights
Local labs are a vital part of clinical trials, offering fast, cost-effective insights into safety and efficacy. But...

Waiver of Consent vs Waiver of Written Consent
One is very common, the other is rarely granted. Learn the key differences between waiver of consent &...

A Digital-First Mindset Shift on eConsent
eConsent plays a pivotal role in optimizing modern clinical trials, but the nuance around its adoption for pharmaceutical...

Preparing Your eConsent Study for IRB Approval
In this blog we cover how eConsent benefits trials and aligns with IRB ethical commitments, how to prepare...

Unlocking the power of Real-World Evidence in FDA submissions
An avalanche of data from so many real-world sources is available today, with the promise of more data...

AKRN optimizes patient assessment collection with Castor ePRO
Castor's ePRO solution enabled AKRN and Company Z to engage with a participant population that, on their own,...

What to look for in an EDC system: a practical guide for clinical trial teams
An electronic data capture (EDC) system is software used to collect, clean, transfer, process, and store data in...

AKRN creates the ideal case report form using Castor EDC
Learn how AKRN and Company Y were able to complete their 100-person study in nine months to help...

AKRN handles mid-study changes in medical device clinical trials with Castor EDC
AKRN Scientific Consulting, a medical device CRO, used Castor's flexible EDC solution to handle constant mid-study changes for...
5 steps for patient engagement and better outcomes in decentralized clinical trials
For successful decentralized clinical trials, researchers must encourage patient engagement. Learn 5 steps that modern companies are taking...

Automated data capture with eSource to train an AI algorithm
Castor’s eSource platform streamlined collecting, capturing & processing large amounts of clinical data to train AusculThing’s AI algorithm.