
Categories
Connect with us on LinkedIn

Video Conferencing for Remote eConsent: 5 Reasons to Make the Switch
Castor is using video conferencing to meet participant needs and lower site burden in clinical trials. Find 5...

How to Prepare an IRB Submission for a Remote eConsent Study
If you are planning on using eConsent in a trial, you’ll need to include it in your IRB...

Understanding the Nuances of Informed Consent in Clinical Trials
Informed consent is a critical aspect of clinical trials. But what criteria must be met? And is there...

Putting PRIDE Into Action at Castor
Castor's Action Hours during Pride Month gave all Castorians the opportunity to educate themselves and celebrate the LGBTQIA+...
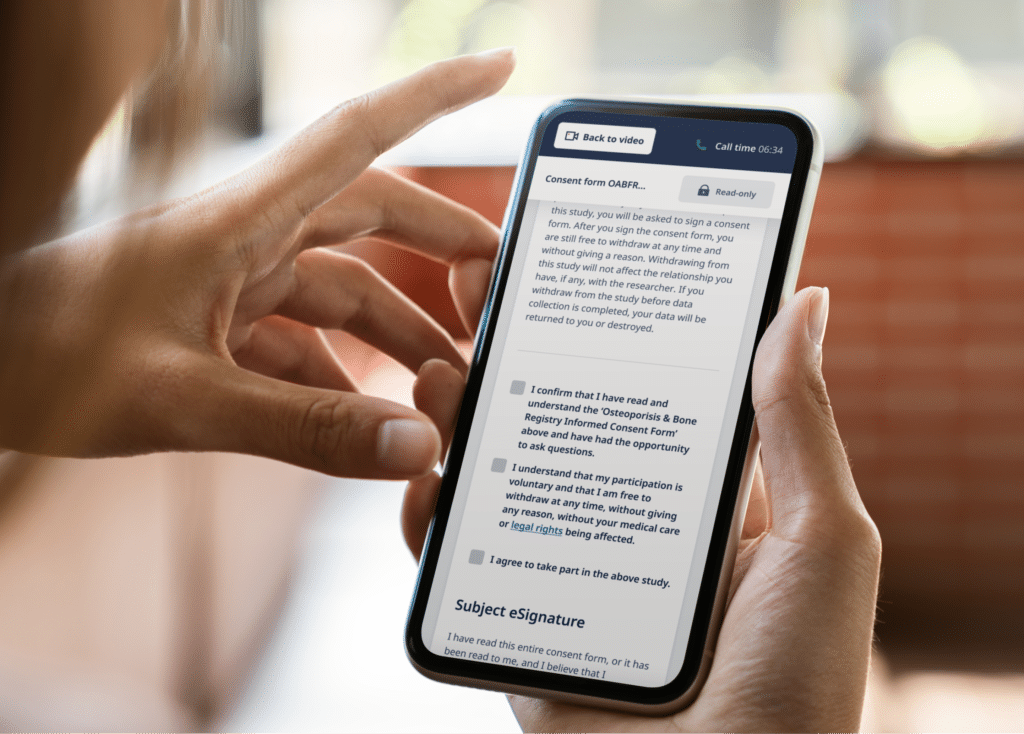
The True Meaning & Value of eConsent in Clinical Trials
The future of informed consent is digital. Learn about the benefits, the process, the regulatory landscape, and the...

Putting Well-Being First: How Castor Promotes a Happy and Healthy Team
May is Mental Health Awareness Month, and Castor initiated a conversation about well-being at work and mental health...

The Ultimate Guide to Post-Market Surveillance: What You Need to Know
In today’s rapidly evolving regulatory landscape, ensuring medical device safety and performance goes beyond the approval phase. Effective...

A day in the life of a Director, Business Development at Castor
We sat down with two of our Business Development Directors to find out what their work involves, why...

Castor Provides Continued Support to the World Health Organization, Powering the World’s Largest Adaptive COVID-19 Trial
Castor announced continued support for the WHO Solidarity trial, the world's largest adaptive COVID-19 trial.

Castor: A Journey From 0–100!
We are reaching the milestone of 100 employees at Castor!

Women in Technology: Castor Interview Series 5 – Marta Ornelas Mauricio
Our Women in Technology series continues with Marta Ornelas Mauricio, QA Tester.

Women in Technology: Castor Interview Series 4 – Alexandra Marinescu
Our Women in Technology series continues with Alexandra Marinescu, Product Owner.

Making a Real-World Difference for Patients & Clinical Trials
Learn why myTomorrows utilizes Castor to monitor treatment and assess benefit/risk its users, connecting patients with expanded trial...

Women in Technology: Castor Interview Series 3 – Luciana Mendonça
Our Women in Technology series continues with Luciana Mendonça, Software Engineer.

Women in Technology: Castor Interview Series 2 – Elena Tanturovska
Our Women in Technology series continues with Elena Tanturovska, QA Automation Engineer.

PMCF & RWE: Roles of Medical Device Registry Studies & ePROs
Registry studies focused on a specific medical device are a valuable tool to capture the safety and outcomes...

Castor Inks Big Pharma Deal As Decentralized Trials Business Continues to Show Exponential Growth
Hoboken, New Jersey: March 10, 2021: Castor, a leading provider of clinical trial technology, today announced the signing...

Women in technology: Castor Interview Series, Roxana Cristian
In the Castor D&I interview series, Castorian women in technology share their experiences and thoughts on making the...