
Categories
Connect with us on LinkedIn
It’s a Matter of Choice: Hybrid eConsent for On-Site or Remote Patient Enrollment
Thought leaders from Castor and Lightship discuss opportunities that will help researchers develop a more successful overall strategy...

eBook: eConsent
Download this eBook to gain a comprehensive overview of eConsent, how it satisfies global regulatory requirements and key...

Digital Therapeutic Cybersecurity: Keeping Participant Data Safe
DTx represent an emerging field of software-driven, evidence-based products intended to “prevent, manage, or treat a medical disease...

How Medical Device Trials Can Benefit from Decentralized Methods
At Castor, we hope to see DCT extend into all trials as we work alongside researchers in the...

8 Data Types to Collect Remotely
Decentralized clinical trials (DCT) are collecting many kinds of data remotely, and this data is only expected to...

What Is eConsent in Clinical Trials?
eConsent accelerates enrollment, increases retention, and reduces site burden. Find out why it’s the way of the future...

Case Study: COVID-RED Uses Castor’s Decentralized eClinical Platform
Download this case study to learn how Castor responded to the disruptive impact of COVID-19 and helped the...

Developing an Infrastructure for Patient-Centric Decentralized Trials
Castor and Lightship discuss the infrastructure needs for providing the best patient experience and ensuring data integrity in...

Three Ways Remote eConsent Improves Trial Efficiency
eConsent accelerates enrollment, increases retention, and reduces site burden. Learn why it’s the way of the future for...

Castor is a Major Contender in Everest Group PEAK Matrix® of Decentralized Clinical Trial Products Assessment 2021
Download the “Decentralized Clinical Trial Products PEAK Matrix®” assessment by Everest Group to learn why Castor is a...

Video Conferencing for Remote eConsent: 5 Reasons to Make the Switch
Castor is using video conferencing to meet participant needs and lower site burden in clinical trials. Find 5...

How to Prepare an IRB Submission for a Remote eConsent Study
If you are planning on using eConsent in a trial, you’ll need to include it in your IRB...

Understanding the Nuances of Informed Consent in Clinical Trials
Informed consent is a critical aspect of clinical trials. But what criteria must be met? And is there...
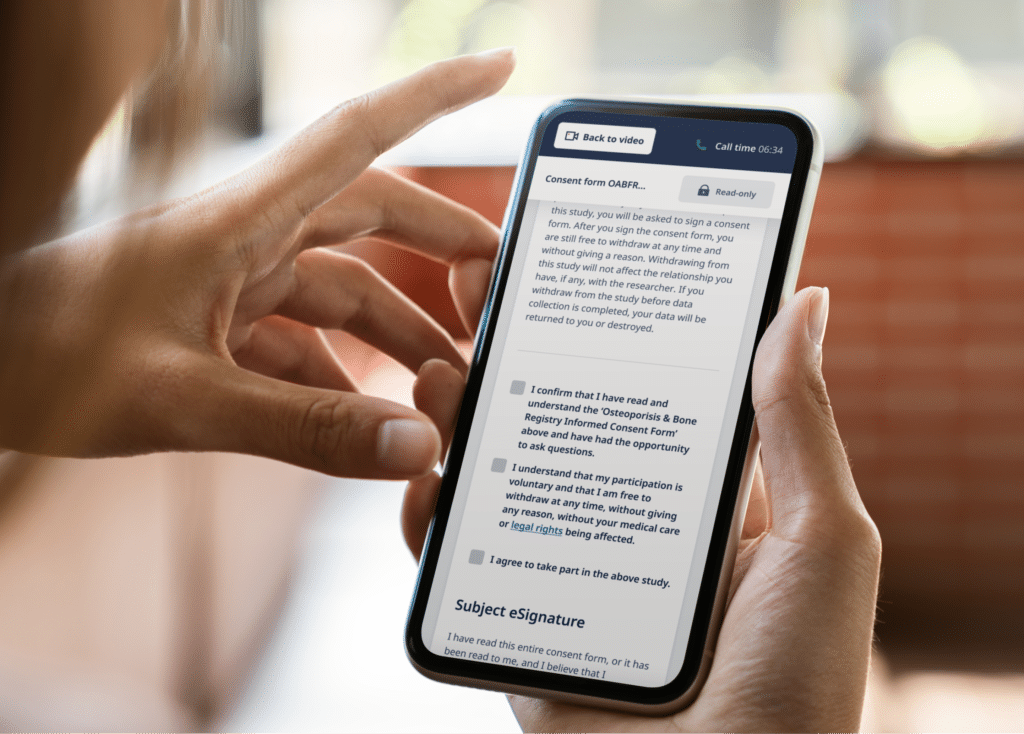
The True Meaning & Value of eConsent in Clinical Trials
The future of informed consent is digital. Learn about the benefits, the process, the regulatory landscape, and the...

Demystifying CDISC standards in the modern era of Decentralized Clinical Trials
Learn key compliance requirements, data collection tips, and important dates for the EU Medical Device Regulation in this...

Castor Provides Continued Support to the World Health Organization, Powering the World’s Largest Adaptive COVID-19 Trial
Castor announced continued support for the WHO Solidarity trial, the world's largest adaptive COVID-19 trial.

Introducing Castor Connect
Watch this webinar for tips on how to ensure research continuity during COVID-19 by running your trial with...

Making a Real-World Difference for Patients & Clinical Trials
Learn why myTomorrows utilizes Castor to monitor treatment and assess benefit/risk its users, connecting patients with expanded trial...