
Categories
Connect with us on LinkedIn

Phase 4 and real-world evidence: not a spectrum, a strategic choice
Phase 4 clinical trials and real-world evidence (RWE) studies serve fundamentally different purposes in post-approval drug development. Phase...

Castor Joins C-Path eCOA Consortium, Surpassing 300 Industry Study Milestone
Castor joins the Critical Path Institute (C-Path) eCOA Consortium, bringing insights from 300+ industry studies to advance participant-centric...

Obesity RWD Compliance: The Direct-to-Patient Guide
Over 2,000 active obesity and GLP-1 studies are competing for the same patients. Most still run on traditional...

Own Your Data, Accelerate Your Cure Castor Catalyst — Rare Disease Insight Brief
Rare disease foundations no longer need to wait for pharmaceutical industry interest to develop treatments for their communities....

Castor’s Global e-Consent Matrix
Electronic consent is now approved or conditionally approved in 27 of 33 countries assessed in Castor's eConsent Country...
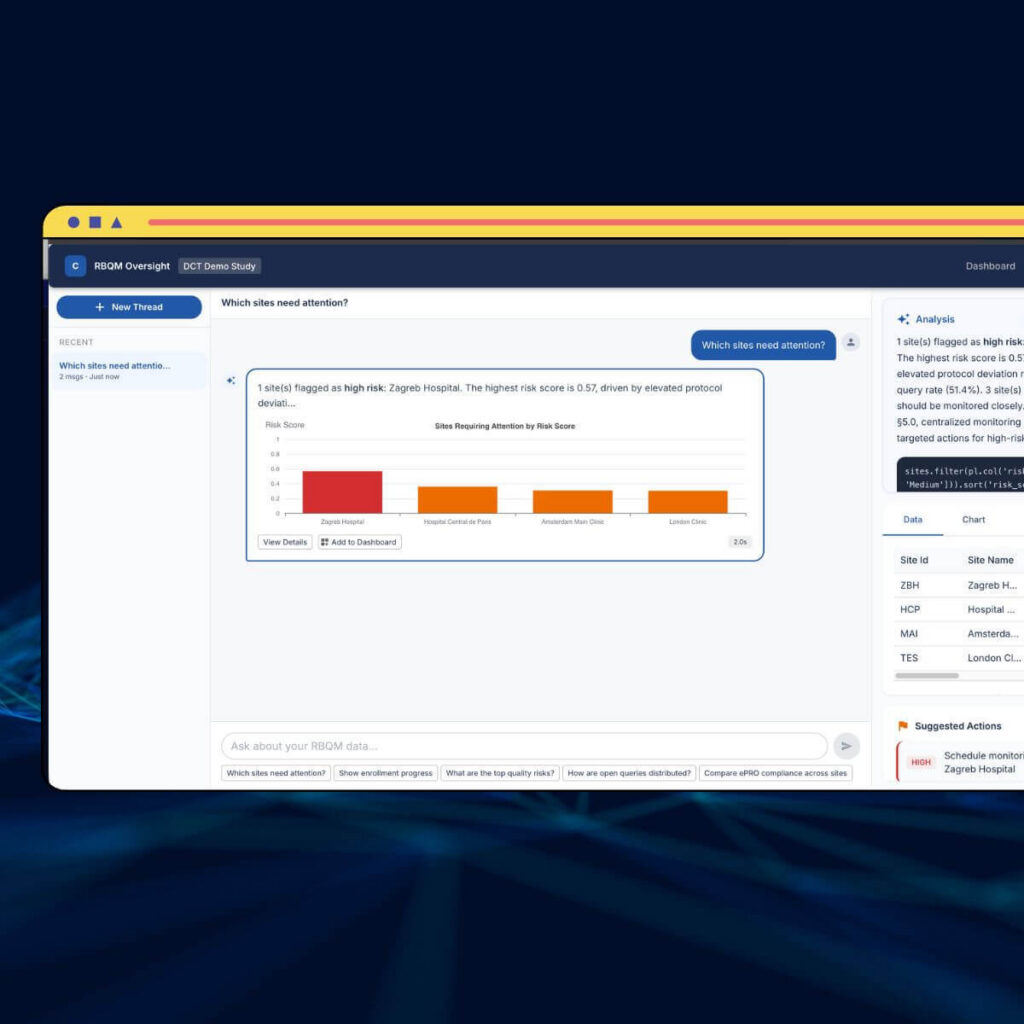
ICH E6(R3) is here: what your centralized monitoring strategy needs right now
ICH E6(R3) is in effect. Castor's recap covers centralized monitoring, sponsor accountability, and closing the biotech RBQM gap.

Practical ICH E6(R3) Oversight for Your Centralized Monitoring Strategy – On Demand
Adopting ICH E6(R3) requires a shift from site-based monitoring to centralized oversight. Watch this on-demand Product Spotlight to...

Castor Professional Services: Expert Clinical Study Builds
Castor’s Professional Services team offers end-to-end support for clinical trial implementation, including EDC build, eCOA, and data management....

The real-time monitoring dilemma
Offline eCOA capability creates regulatory tension with EMA device-as-source requirements while eliminating real-time monitoring capabilities that drive 80-95%...

SCOPE 2026 Meet us at Booth #1612
February 2nd – 5th | Rosen Shingle Creek in Orlando, Florida
Join Castor at SCOPE US 2026, February 2-5 at Orlando's Rosen Shingle Creek. Discover how our "self-driving" clinical...

Executing Concurrent Decentralized RCTs and RWE at Scale
Discover how Sword Health used Castor to validate digital MSK care with scalable RCTs and real-world evidence strategies.

3-Week Database Builds That Withstood FDA Review
Castor’s EDC enabled Gameto’s 3-person team launch in 3 weeks, run 9 IVF studies on 4 continents, and...

Product Spotlight The Self-Driving Study with Castor Catalyst – On Demand
Join Castor CEO Derk Arts to explore Castor Catalyst, the agentic AI platform built with Google Cloud AI....

Is Your eCOA UAT Stuck in Time?
Modern clinical trials increasingly rely on complex, time-sensitive logic within eCOA systems, such as narrow compliance windows and...

The End of the “PRO Tax”: Top 10 Commercial PROs & their cost-effective alternatives
The clinical trial industry faces a "PRO Tax"—the high costs and operational delays associated with licensing and implementing...

The Silent Saboteurs: Why Rater Drift and Site Unpreparedness Cost CNS Trials More
CNS trials fail not from technology limitations but organizational factors. Research reveals 55% of sites lack adequate eCOA...

Hospital-Based eCOA Implementation: Real Challenges in Infectious Disease Trials
Hospital-based infectious disease trials face unique eCOA implementation challenges including IT security delays, staff turnover, and patient acuity...

Implications of Assessing Overall Survival in Oncology Studies
The FDA’s August 2025 draft guidance reshapes oncology clinical trials by requiring pre-specified overall survival (OS) analysis in...