
Categories
Connect with us on LinkedIn

The future of rare disease research
Each rare disease research breakthrough offers the promise of a longer, better life for patients who otherwise may...

What patient experience can learn from the 7 Principles of Universal Design | White paper
Castor explores how clinical researchers improve the participant experience by having good user design in their patient-facing technology

Getting Started with DCT in 2022
Castor advisor Craig Lipset, suggests that you consider DCT technology as three separate tiers. Learn more about these...
It’s a Matter of Choice: Hybrid eConsent for On-Site or Remote Patient Enrollment
Thought leaders from Castor and Lightship discuss opportunities that will help researchers develop a more successful overall strategy...

eBook: eConsent
Download this eBook to gain a comprehensive overview of eConsent, how it satisfies global regulatory requirements and key...

3 Ways eConsent Tackles the Challenges of Modern Clinical Trials
Although eConsent struggled to gain momentum and wider acceptance pre-pandemic, it's now a major part of clinical research....

Trialbee and Castor Partner to Democratize Access and Simplify Enrollment to Clinical Trials Globally
Trialbee, the leading enrollment performance company, and Castor, a leading provider of decentralized and hybrid clinical trial solutions,...

The evolution of eConsent
Since it was first instituted in the 1960s, written consent to participate in research studies has undergone very...

What Is eConsent in Clinical Trials?
eConsent accelerates enrollment, increases retention, and reduces site burden. Find out why it’s the way of the future...

Castor Leads Next-Gen Clinical Trials that Support Digital Therapeutics
Castor is supporting the next generation of digital therapeutics that can improve patient lives with disease-modifying potential.

Three Ways Remote eConsent Improves Trial Efficiency
eConsent accelerates enrollment, increases retention, and reduces site burden. Learn why it’s the way of the future for...

eConsent Readiness in 12 Countries
eConsent is imperative for continuing research during the COVID-19 pandemic and beyond, but regulations can vary widely across...

Castor Launches Scalable Patient Enrollment and eConsent Platform to Increase Patient Access and Reduce Recruitment Timelines
Castor's new eConsent platform will improve remote patient recruitment and enrollment as well as access to diverse populations...

Video Conferencing for Remote eConsent: 5 Reasons to Make the Switch
Castor is using video conferencing to meet participant needs and lower site burden in clinical trials. Find 5...

How to Prepare an IRB Submission for a Remote eConsent Study
If you are planning on using eConsent in a trial, you’ll need to include it in your IRB...

Understanding the Nuances of Informed Consent in Clinical Trials
Informed consent is a critical aspect of clinical trials. But what criteria must be met? And is there...
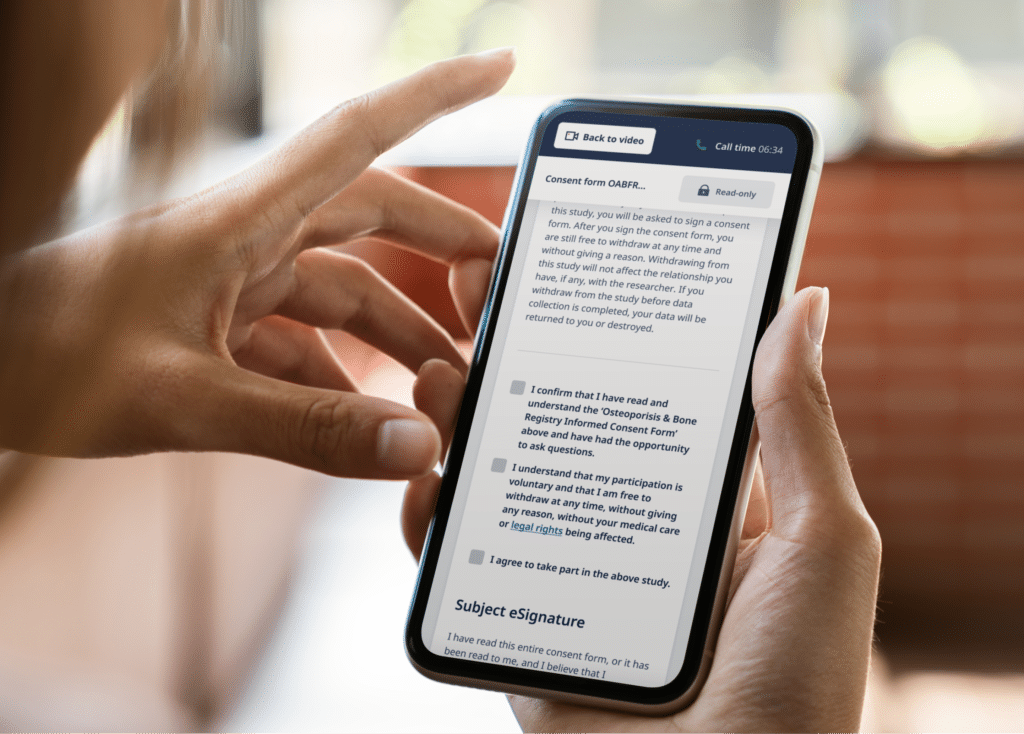
The True Meaning & Value of eConsent in Clinical Trials
The future of informed consent is digital. Learn about the benefits, the process, the regulatory landscape, and the...

Castor Inks Big Pharma Deal As Decentralized Trials Business Continues to Show Exponential Growth
Hoboken, New Jersey: March 10, 2021: Castor, a leading provider of clinical trial technology, today announced the signing...