
Categories
Connect with us on LinkedIn

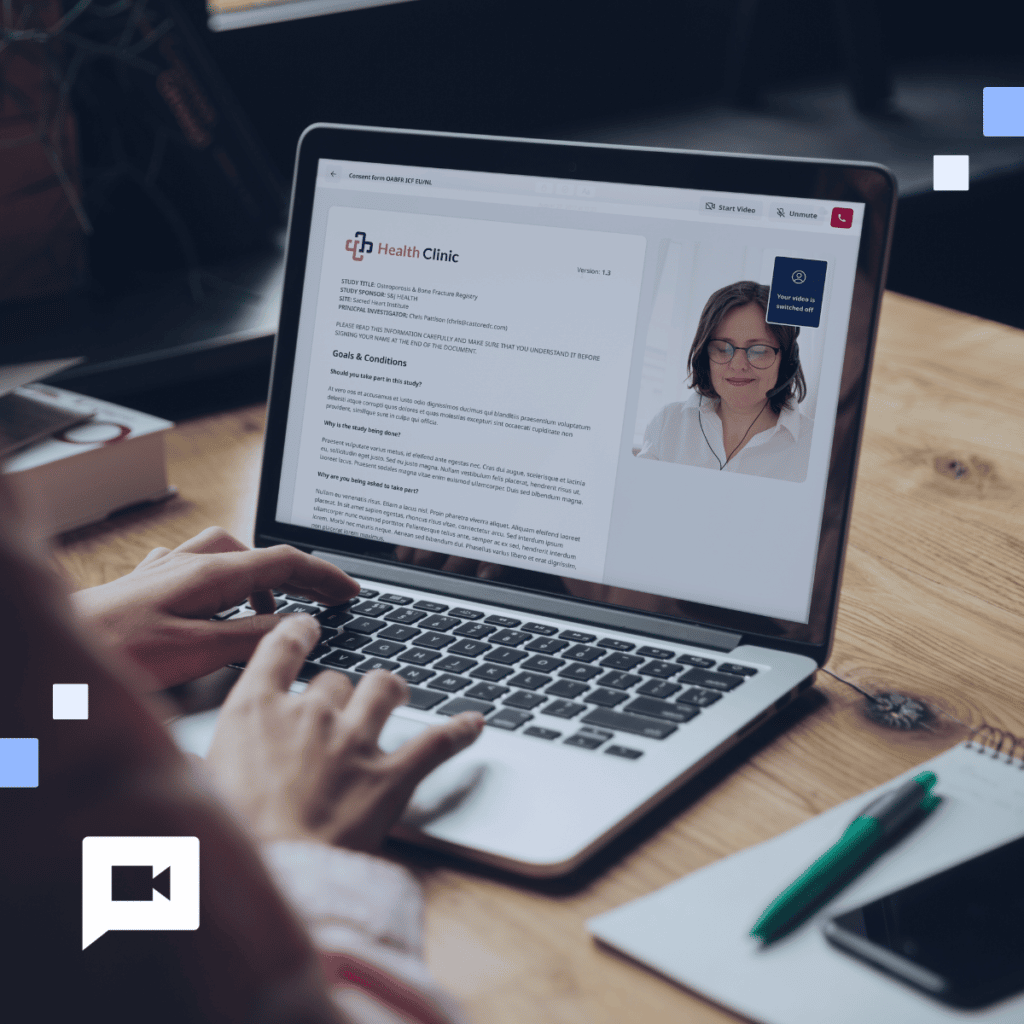
Unlocking the potential of GPT-generated eConsent for IRB approval
As the adoption of real-world evidence (RWE) in healthcare continues to grow, and with the FDA embracing these...

Unlocking the power of Real-World Evidence in FDA submissions
An avalanche of data from so many real-world sources is available today, with the promise of more data...

The role of patient-centric technologies & tokenization in RWE studies
In this on-demand webinar we join forces with UBC & Datavant to dive deep into patient-centric technologies &...

Considerations for the use of RWE in FDA Assessments
As the adoption of real-world evidence (RWE) in healthcare continues to grow, and with the FDA embracing these...

eConsent Readiness Guide in 18 Countries

Improving Your Consent Process with Generative AI
As the adoption of real-world evidence (RWE) in healthcare continues to grow, and with the FDA embracing these...

Real-World Evidence: The Importance of Standardization & Democratization
As the adoption of real-world evidence (RWE) in healthcare continues to grow, and with the FDA embracing these...

Castor announces appointment of Brad Hightower, leading clinical trials executive, to its advisory board
Castor announces the appointment of Brad Hightower, a leading clinical trials executive, to its advisory board.

2023 Making a Positive Impact
As we kick-off 2023 with a refreshed mindset, we take inventory of what is important and focus on...

Making Every Data Point Count: The Value of Machine-Readable, FAIR (Meta)data in Clinical Research
Martijn Kersloot, PhD, Product Owner at Castor, gives an introduction into the FAIR Data Principles and highlights why...

3 ways to unlock the value of real-world evidence (RWE)
Learn best practices on RWE, Castor's offerings for collecting RWD, and 3 different RWE case studies from Castor...

2023 Guide to Clinical Data Collection under the EU Medical Device Regulation (EU MDR)
Learn key compliance requirements, data collection tips, and important dates for the EU Medical Device Regulation in this...

AKRN optimizes patient assessment collection with Castor ePRO
Castor's ePRO solution enabled AKRN and Company Z to engage with a participant population that, on their own,...

What to look for in an EDC system: a practical guide for clinical trial teams
An electronic data capture (EDC) system is software used to collect, clean, transfer, process, and store data in...

Digital Trials: Reinventing new definitions for clinical research
There are ongoing debates about whether or not our industry should call all decentralized ones digital trials; however,...

Castor Launches Real-World Evidence Offering to Simplify Post-Marketing Clinical Trials
Real-world evidence has the potential to transform evidence based medicine, by generating a body of evidence that provides...

Castor Expands Platform Capabilities, Releases Virtual Visit Feature to Optimize the Clinical Trial Experience for Researchers and Patients
By reducing the effort, cost, and time needed, Castor Virtual Visits enhance convenience and flexibility - upgrading the...
What Patient Experience Can Learn from the 7 Principles of Universal Design
Learn why having a good user interface is crucial for enrolling and retaining patients in a decentralized clinical...