
Categories
Connect with us on LinkedIn

Castor Research Award – Nominee #1
Back in December, we announced that at the end of 2018 we would be awarding a €3000 prize to the...
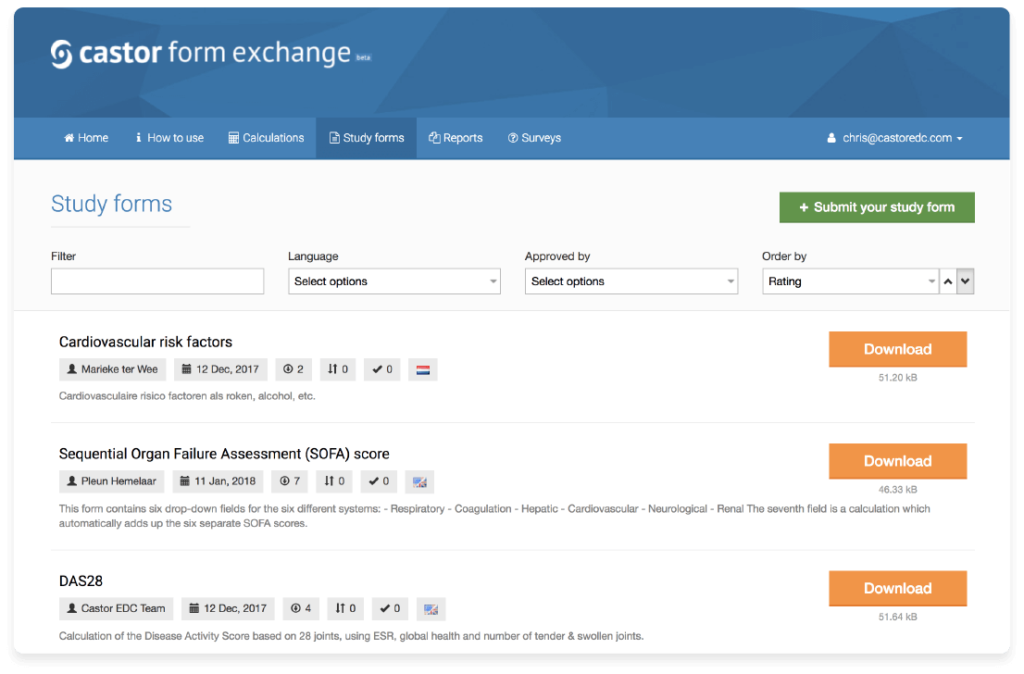
Everything you need to know about Castor’s improved Form Exchange!
The beginning of 2018 has been busy for Castor; this quarter, we have been working on various new...
The countdown to GDPR begins!
Castor is getting ready for GDPR coming up this May! What is GDPR? The General Data Protection Regulation...

Multinational Studies with Castor EDC (HIPAA & GDPR Compliance)
Since the beginning of our journey, we have made it our mission to accelerate medical research by unlocking...

Award-winning ‘So Hip’ study collected data using Castor
The prize for the best research at the Amsterdam University of Applied Sciences in 2017 went to Margriet...

Researcher 2 Researcher Program
At Castor, our aim is to help researchers who are looking for ways to increase the quality of...

How we’re gearing up for GDPR
In the past two decades, technology has drastically changed how we capture, use, and store data. As a...

Introducing: The Castor Research Award
Are you using Castor for medical research? As of 2018, we will be awarding the most promising research...

Metadata: what are they and why do I need them?
In the last couple of years, researchers have been encouraged to start using metadata when they collect data...

Net Promoter Score (NPS) – Would you recommend Castor to colleagues?
In the last two months, you might have noticed a small pop-up at the bottom of your Castor...
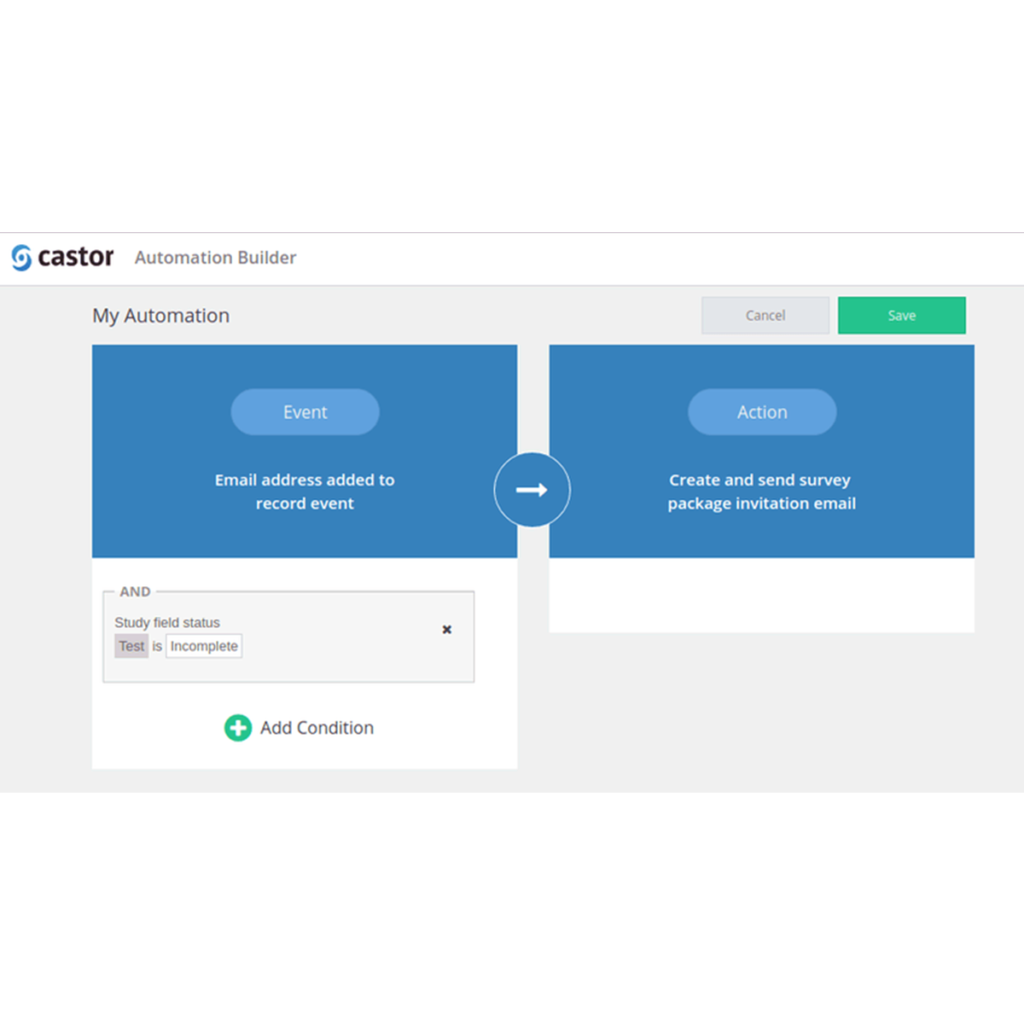
The Automation Engine is now available as a beta feature!
As part of the 2017.6 release, the automation engine has been added as a beta feature! You can...

Innovative Cardiology Crowdfunding: Fast Security for Your Heart
Sharing photos, ordering groceries, online dating. It seems like there’s a mobile application for everything. With millions of...

Our €1.1M grant and how it will change medical research
As you may have seen in recent articles published on Tech.eu, EU Startups, or Nu.nl; Castor was recently awarded...

Keep Track of Your Study More Easily With Ldot
With large quantities of patients, extensive calling lists, SMS reminders and more, keeping track of a research project...

What’s in the Footer? A Quick Guide to Software Versioning & How Castor Does It
Very recently a new item was placed in the footer of Castor: the version number of the software!...
ePRO: the electronic solution for patient reported outcomes – OLD
Traditionally done on paper, surveys are a common way to collect data from study participants. Surveys are questionnaires...
How we chose the winner for our survey using R
Some time ago we ran a survey to find out why people had stopped using Castor EDC. To improve...

How do you perform GCP-compliant research with Castor EDC?
Castor EDC allows researchers to collect and manage data in accordance with the Good Clinical Practice (GCP) guidelines...