
Categories
Connect with us on LinkedIn

Castor Launches Scalable Patient Enrollment and eConsent Platform to Increase Patient Access and Reduce Recruitment Timelines
Castor's new eConsent platform will improve remote patient recruitment and enrollment as well as access to diverse populations...

Video Conferencing for Remote eConsent: 5 Reasons to Make the Switch
Castor is using video conferencing to meet participant needs and lower site burden in clinical trials. Find 5...

How to Prepare an IRB Submission for a Remote eConsent Study
If you are planning on using eConsent in a trial, you’ll need to include it in your IRB...

Castor Raises $45M Series B to Modernize the Clinical Trial Process and Maximize the Impact of Research Data on Patient Lives
Castor, a leading provider of clinical trial software, announced today the closing of a $45 million Series B...

Understanding the Nuances of Informed Consent in Clinical Trials
Informed consent is a critical aspect of clinical trials. But what criteria must be met? And is there...

Putting PRIDE Into Action at Castor
Castor's Action Hours during Pride Month gave all Castorians the opportunity to educate themselves and celebrate the LGBTQIA+...
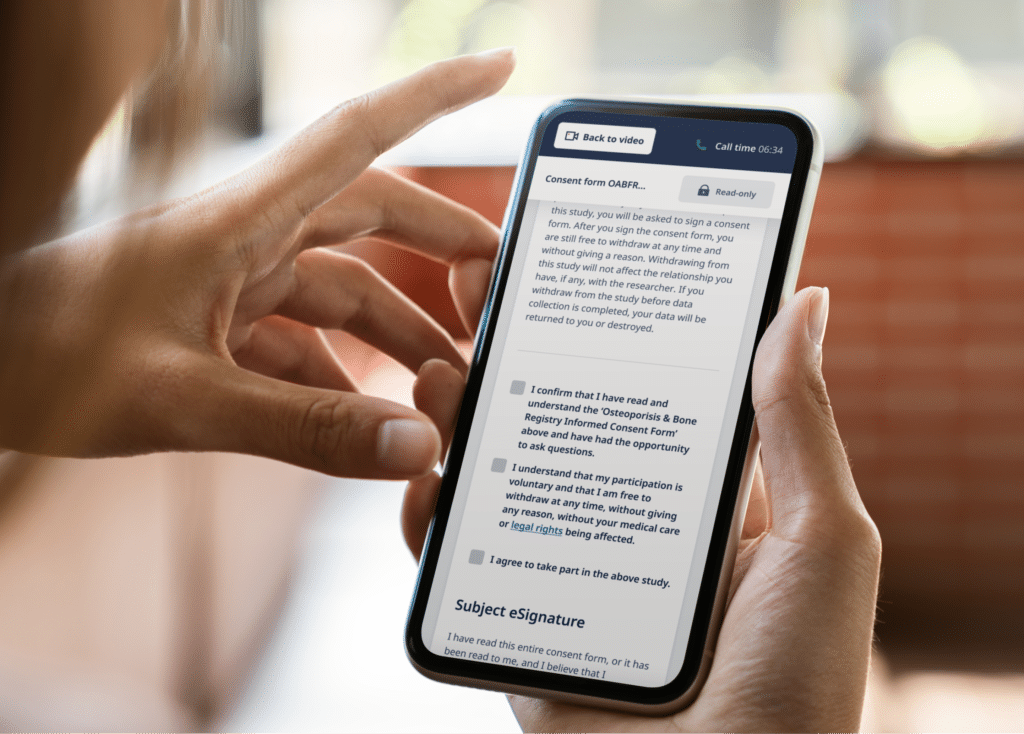
The True Meaning & Value of eConsent in Clinical Trials
The future of informed consent is digital. Learn about the benefits, the process, the regulatory landscape, and the...

Lightship Announces Partnership with Castor to Scale Virtual Clinical Trials Worldwide
Lightship and Castor, leading providers of decentralized clinical trial solutions, today announced a strategic partnership to run direct-to-patient...

Demystifying CDISC standards in the modern era of Decentralized Clinical Trials
Learn key compliance requirements, data collection tips, and important dates for the EU Medical Device Regulation in this...

Putting Well-Being First: How Castor Promotes a Happy and Healthy Team
May is Mental Health Awareness Month, and Castor initiated a conversation about well-being at work and mental health...

Castor Continues Rapid Growth and Momentum Powering COVID-19 Studies with Global Decentralized Clinical Trial Platform
Castor is supporting more than 250 COVID-19 studies in 40 countries across 1,750 hospitals as it continues to...

The Ultimate Guide to Post-Market Surveillance: What You Need to Know
In today’s rapidly evolving regulatory landscape, ensuring medical device safety and performance goes beyond the approval phase. Effective...

A day in the life of a Director, Business Development at Castor
We sat down with two of our Business Development Directors to find out what their work involves, why...

Introducing FormSync: Castor’s Solution to Mid-Study Changes
Join this webinar and learn how to easily change, review and approve eCRF forms in your test environment,...
Essential Guide to Post-Market Clinical Follow-Up (PMCF) for Medical Devices
Learn the full impact of PMCF under the EU MDR and how to best prepare your medical device...

Castor Provides Continued Support to the World Health Organization, Powering the World’s Largest Adaptive COVID-19 Trial
Castor announced continued support for the WHO Solidarity trial, the world's largest adaptive COVID-19 trial.

Castor: A Journey From 0–100!
We are reaching the milestone of 100 employees at Castor!

Introducing Castor Connect
Watch this webinar for tips on how to ensure research continuity during COVID-19 by running your trial with...